- Book Now
-
-
Surgical
- Before & After
-
-
About Surgery
- For Women
- For Men
- Extreme Makeover
- Frequently Asked Questions & Reviews
-
-
-
-
Non-Surgical
-
-
Skin Tightening
- Ultherapy
- FaceTite
- Non-Surgical Facelift
- MyEllevate Neck Lift
- Nécolleté™
- View All
-
-
-
Body Sculpting
- Non-Surgical BBL
-
- Skincare
- Before & After
-
- All Treatments
-
-
Story & Staff
- Our Mission
- Our Staff
- Our Approach
- Meet Dr. Pearlman
-
- Before & After
- Contact
- Blog
Welcome to Pearlman Aesthetic Surgery for Kybella in NYC on Park Avenue.
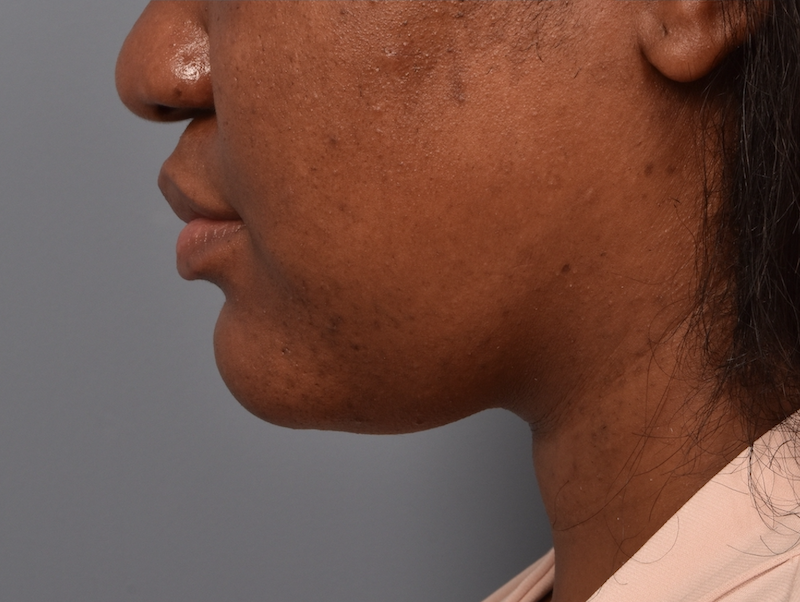
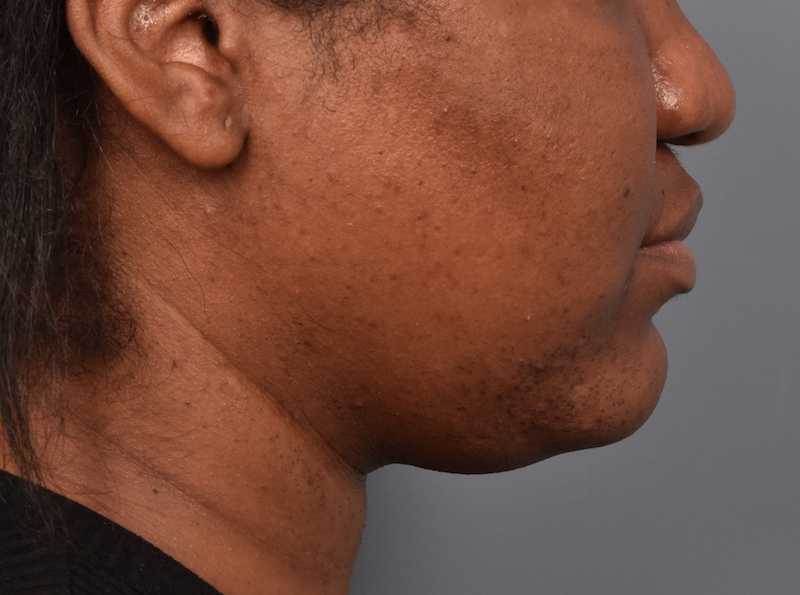
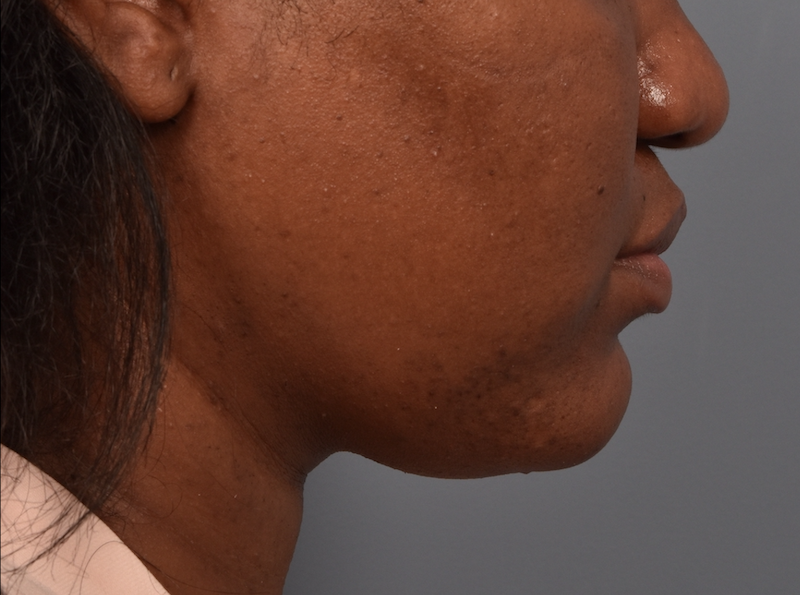
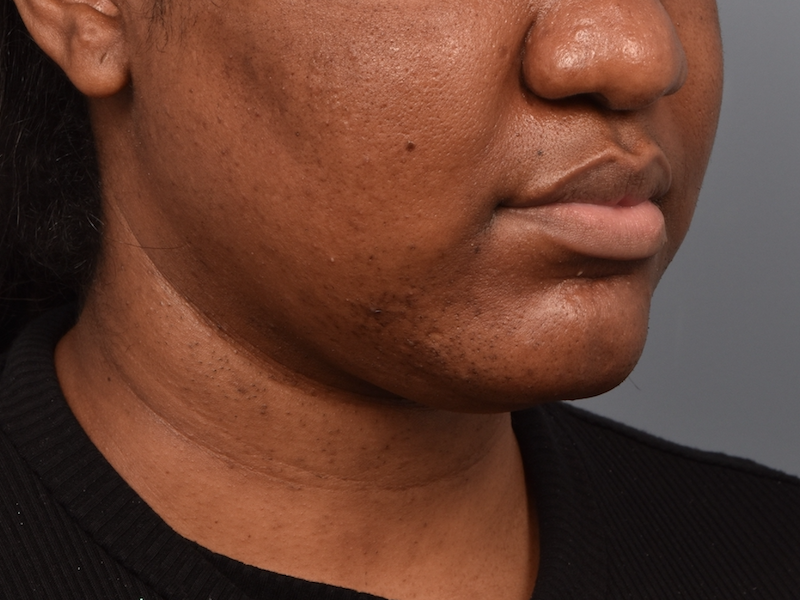
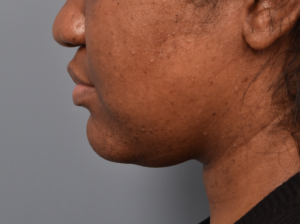
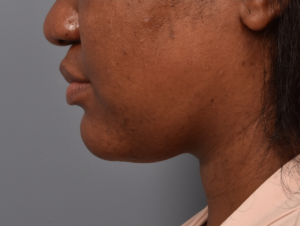
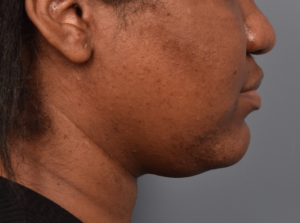
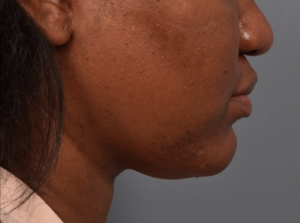
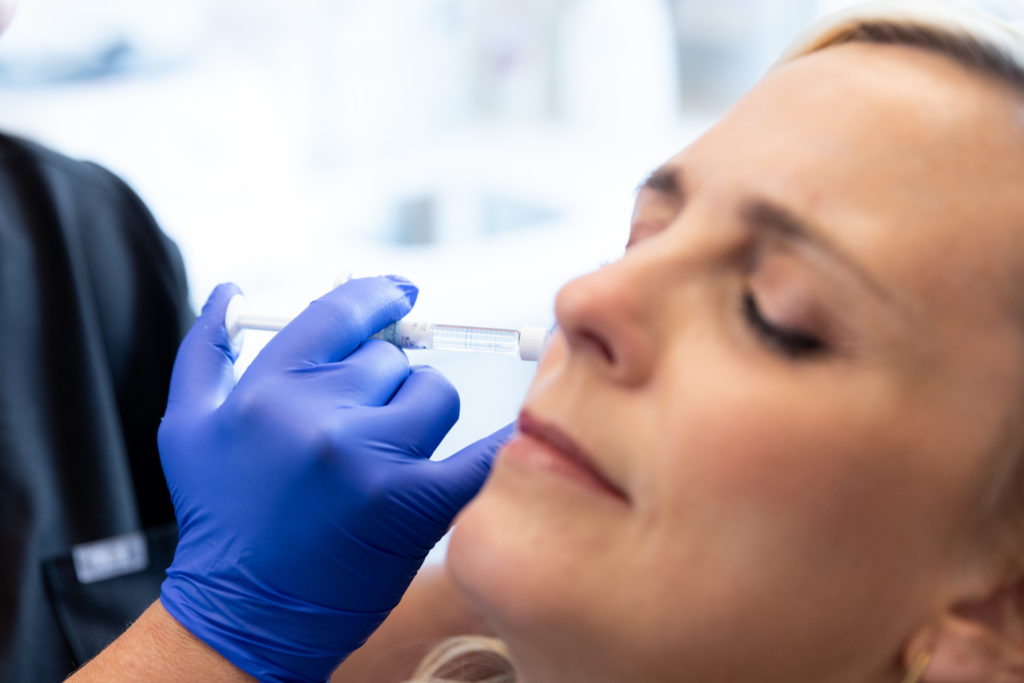
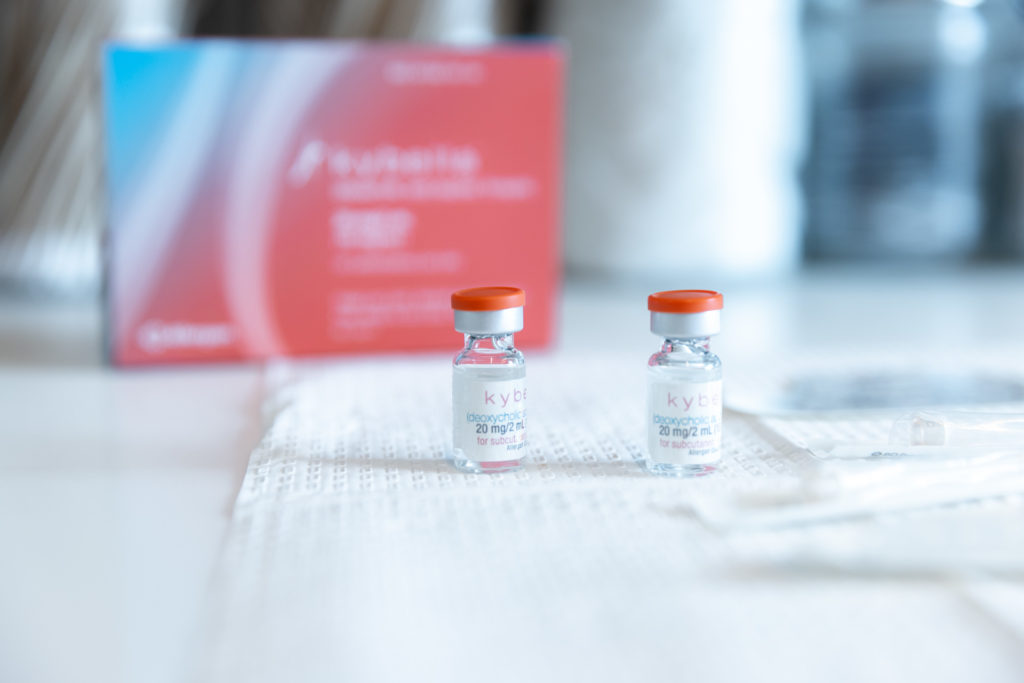
Kybella® is the ideal FDA-approved procedure to permanently reduce a double chin and enhance jawline contouring without surgeries, incisions, or recovery period. Our providers are some of the first and foremost practitioners of the Kybella treatment in the U.S., having received specialized training from Kythera Biopharmaceuticals.
A slender, youthful appearance.
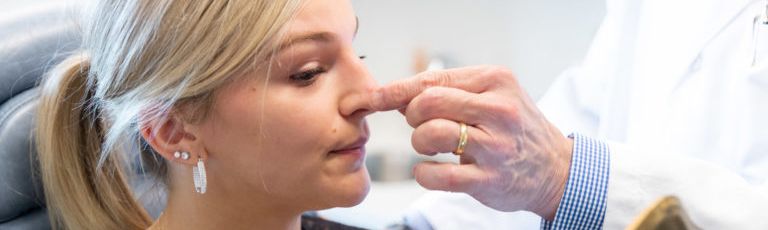
Do you want to get rid of your dreaded double-chin? Whether you’re genetically predisposed to formation of a double chin or you struggle losing fat in the area, a targeted double chin treatment can help reduce fat in the neck area.
Kybella is one of the first effective nonsurgical treatment options for reducing submental fullness.
Before this treatment, your only options were surgical — liposuction, platysmaplasty, neck lifts, and chin implants. While surgical procedures are also effective, they lead to a considerable recovery period. You can contour and streamline your chin to project a sleek, slender, and youthful appearance with Kybella in NYC at Pearlman Aesthetic Surgery. Kybella injections are delivered into the fat tissues under the chin.
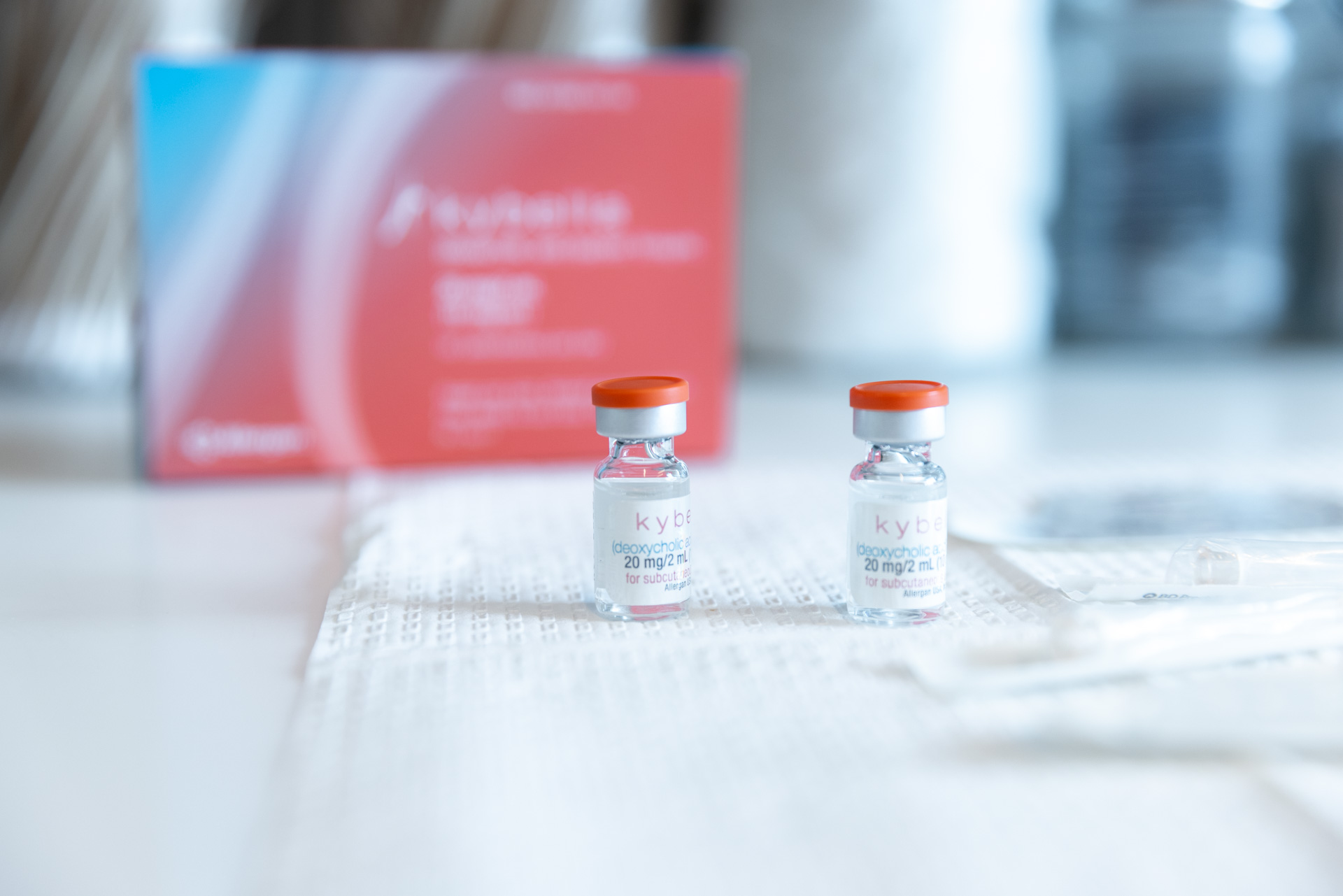
The injection is composed of deoxycholic acid, a naturally occurring substance readily found in the human body, responsible for breaking down and absorbing dietary fat. When we inject Kybella into the submental (chin) area, the synthesized deoxycholic acid dissolves the fat tissues in your double chin, resulting in a streamlined, slender, and contoured chin and jawline. Kybella is an incredibly safe and effective treatment — it has been carefully developed over 9 years, featured in over 20 clinical studies, and used to successfully treat thousands of patients with brilliant results.
Treatment Details
Treatment Duration:
20 to 30 minutes
Downtime:
1 week
Duration of Results:
Permanent, depending on your diet and exercise
See results in:
2 to 4 sessions
Performed By:
Aesthetic Nurse
Anesthetic:
None
Kybella in NYC can treat
Double chin
Excessive adipose tissue
The Prevalence of Submental Fat (Double Chin)
According to a 2015 study by the American Society for Dermatologic Surgery, around 67% of all people report self-consciousness due to a double chin, since it makes you look heavier or older. Submental fullness occurs because of three primary factors:
Genetics: Some people are more predisposed to submental fullness than others.
Age: The likelihood of developing a double chin increases with age.
Weight: You’re more likely to develop a double chin as you gain weight.
Consultation
How it Works
Kybella Consultation
What To Expect
Recovery
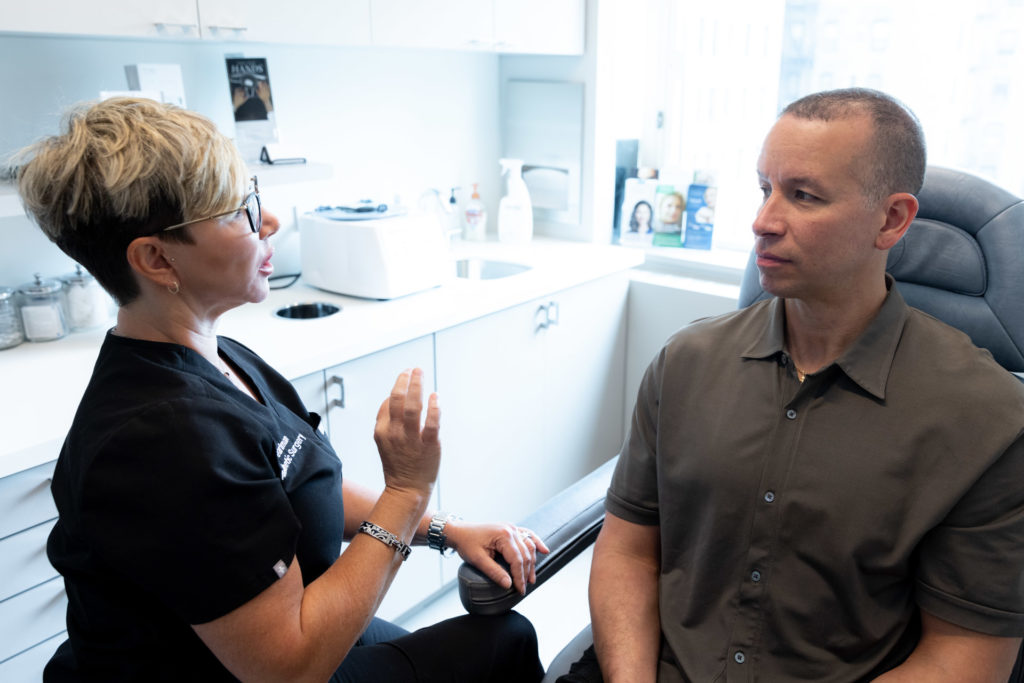
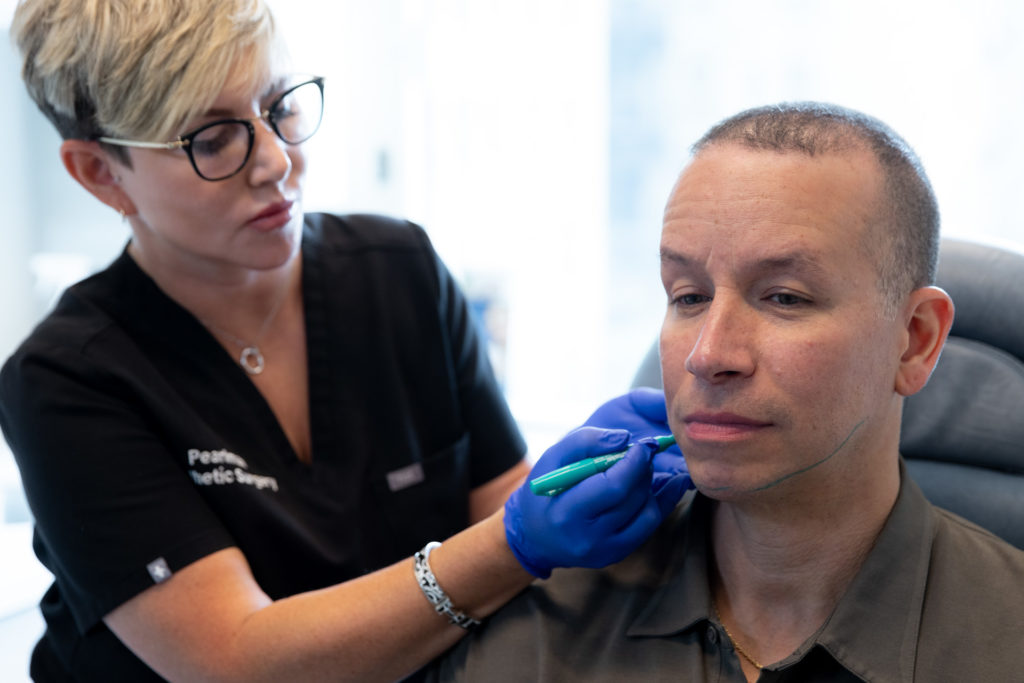
During your consultation for Kybella at our office on Park Avenue, your provider will go over all of your submental treatment options, including Kybella and other surgical options. They will highlight the pros and cons of all possible procedures, discuss your candidature for different treatments, and provide all the necessary information to help you make an informed decision.
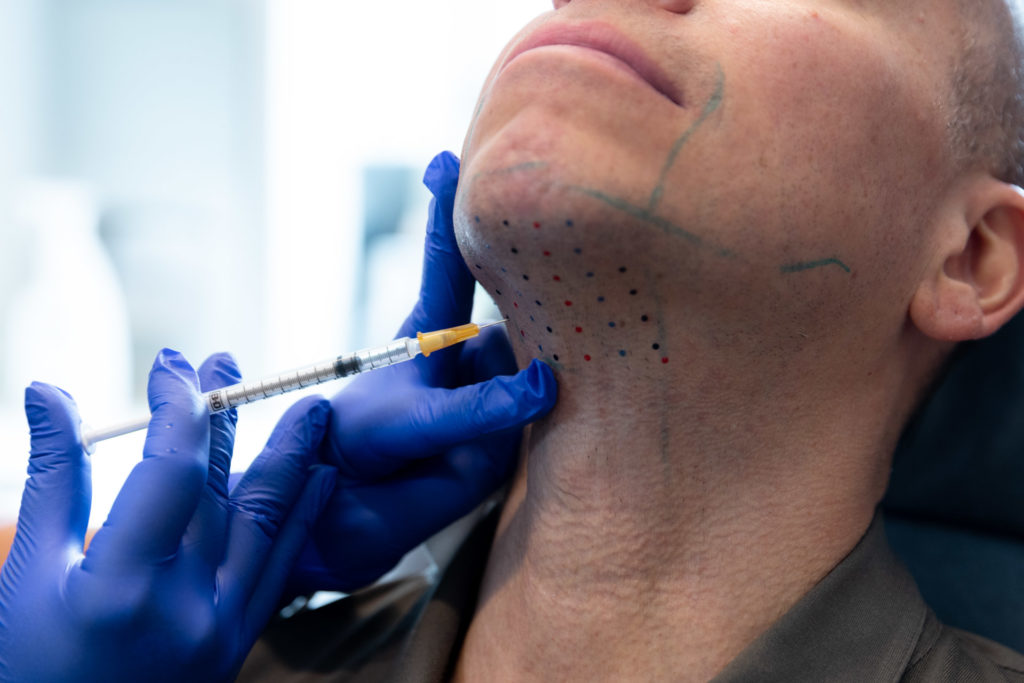
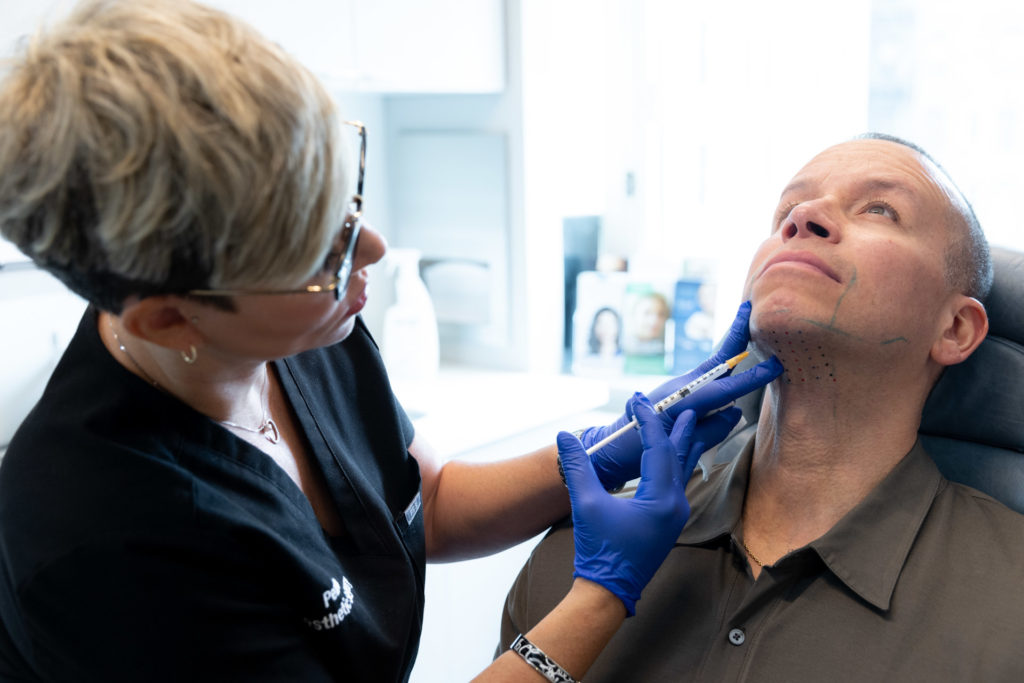
There is no anesthetic needed for this treatment. Your provider will ensure you are comfortable while they inject Kybella into the predetermined treatment areas. The entire process will take between 20 to 30 minutes. Based on your individual treatment plan, you may come to our Manhattan office for 2 to 4 treatments for the optimal results.
Like all injectable treatments, you should expect some bruising and swelling for a few days after your treatment with Kybella in NYC. You can conceal the bruising with some light makeup as soon as the next day.
The injection sites will appear swollen for 4 to 7 days. While you can resume work and most daily activities, you’re advised to avoid strenuous activities, such as working out or lifting weights during this time.
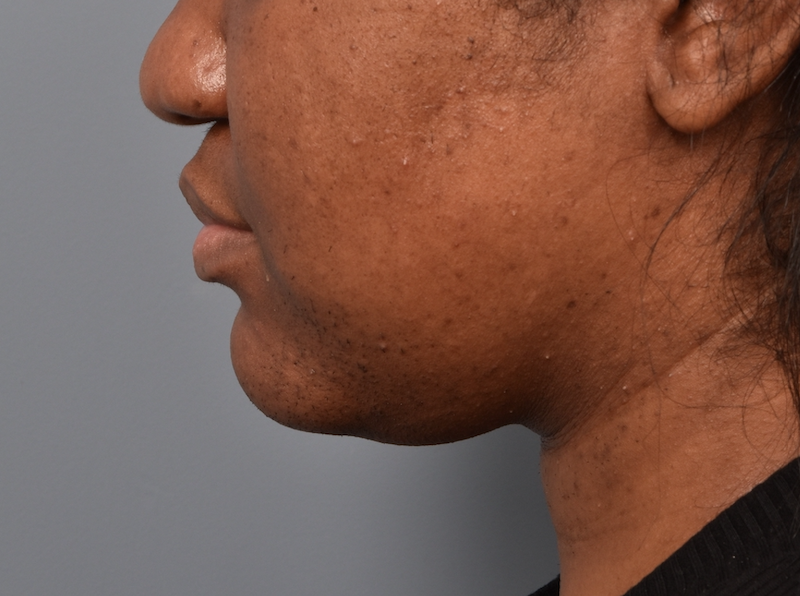
You’ll enjoy newfound confidence with a slimmer neck, contouring jawline, and reduced double chin with a treatment for Kybella in NYC. View our real patient results from this non-surgical procedure.
Frequently Asked Questions
Kybella
Are there are contraindications for Kybella®?
Our providers carefully evaluate each patient to ensure they’re ideally suited for the procedure. Generally speaking, Kybella may not be suitable for patients who:
- Have undergone plastic surgeries in the neck, chin, or jawline.
- Have medical conditions that affect the facial or submental regions.
- Have bleeding problems or swallowing issues.
How long do Kybella® results last?
According to clinical studies, the results from a Kybella treatment are long-lasting, provided there aren’t any drastic lifestyle changes.
How many Kybella® treatments will I need to see optimal results?
The total number of Kybella treatments you will need for optimal results depends on several complex factors. However, most patients need 2 to 6 Kybella injections between intervals of 4 to 6 weeks each. During your consultation for Kybella in NYC at our Manhattan office, Dr. Pearlman will curate the ideal treatment plan for your specific needs based on your age, facial anatomy, cosmetic goals, medical history, and physiology.
How soon will I see Kybella® results?
Generally speaking, Kybella injections dissolve the fat tissues within 10 to 15 minutes after the treatment. However, you may experience swelling due to your body’s natural inflammatory response. You’ll notice the results of your treatment with Kybella in NYC as the swelling subsides after 3 to 4 weeks. Your results will first become evident in 4 to 6 weeks, and you’ll continue seeing gradual improvements through the subsequent treatments. However, please note that everyone’s body is different, and the timeline may be different for you.
Is Kybella® safe?
Kybella injections had similar side effects as other injectable treatments, i.e., swelling, bruising, redness, temporary numbness etc. The treatment was deemed so safe and effective that it was approved by every single member of the FDA Medical Advisory Committee. However, it’s worth noting that improper injection could injure the surrounding skin tissues, such as the regions around your mouth. But that only happens due to improper injection outside the approved region, i.e., the submental region.
What happens if I gain weight after the Kybella® treatment?
Patients in the clinical studies who experienced weight gain after their Kybella® treatment did not see the return of their submental fullness. However, we will have a better understanding of the impact of subsequent weight gain in the future when data becomes available from a broader range of patients who receive this double chin treatment.
Patient
Experiences
Persistent in the Pursuit of Perfection
For decades, Dr. Pearlman has been persistent in his search for perfection for creating the best plastic surgery office in NYC. His leadership brings evolution, experience, expertise, and uniqueness to Kybella patients in our Park Avenue office. The goal of Pearlman Aesthetic Surgery is to help patients to actualize their ideal self-image for the highest level of personal authenticity and attractiveness possible. Our providers couple an attentive bedside manner and genuine concern for our patients’ welfare to deliver the best Kybella results.

Learn if you're a candidate today.
Designed by our aesthetic and surgical specialists, our procedure match tool will help you learn more about the possibilities for your treatment with Kybella in NYC.
Request Appointment
Address
110 E 60th ST #908 NEW YORK, NY 10022
Hours
Monday
9 am – 5 pm
Tuesday
9 am – 5 pm
Wednesday
10 am – 6 pm
Thursday
9 am – 5 pm
Friday
9 am – 5 pm
Sat - Sun
closed
Further Your Results
A variety of other procedures can be combined with Kybella to provide facial contouring. We offer many surgical and non-surgical procedures to achieve this goal that can pair with Kybella in NYC, including: